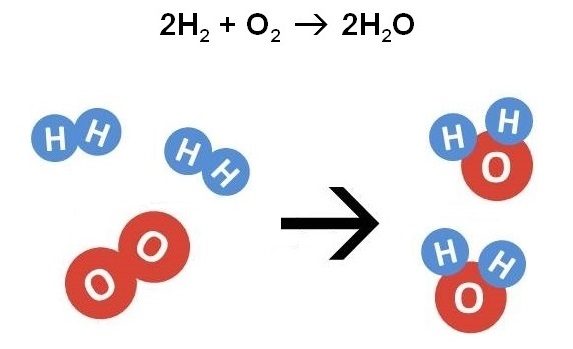
In today’s CRotD, Mr. A burned a balloon full of hydrogen. It didn’t go exactly as planned. Nevertheless, the reaction was a great example to use for yesterday’s topic: chemical reaction equations and how to draw before-and-after pictures.
During the reaction, two H2 molecules split because of the added heat, and when the atoms reformed we were left with pure H2O. Who knew fire could make water?
We used this example to introduce the three main types of chemical reactions. Synthesis is where molecules are put together. Decomposition is where molecules are broken down. And replacement is where elements are simply switched around. In the example above, you are building water molecules out of three smaller pieces, so it is classified as a synthesis reaction. After discussing this example, students practiced classifying reactions and drawing the corresponding pictures.