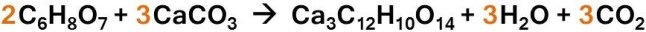Today was the second day of the Marooned on Mars project and most groups began with the very first goal on their list: Obtaining safe drinking water. But this was no easy task. Because we were unable to create H2O using a chemical reaction (Mr. A has learned his lesson about trying to burn hydrogen), we had to resort to trying to purify wastewater.
This was really a three step process. First, we had to lower the PH of the wastewater by adding an acid. Second, we had to filter out any poisons using charcoal. And third, depending on how we viewed the likelihood of germ contamination, we had to boil the water to sanitize it.
Remember everyone, at the end of the week you must hand in a lab procedure that includes a goal, a materials list, a procedure, a chemical reaction equation (if one was used), and a drawing of the reaction. The equation for the wastewater reaction, “citric acid + calcium carbonate,” can be found below.