A few hundred years ago, things were different. In order to light a fire, you would have to get out your flint and your steel, strike them, and hope that the few sparks you made would light your kindling on fire. Today, you simply use a match. A few hundred years ago, they thought that fire would actually destroy the firewood and only the ashes would be left. Today we know better.
Fire cannot destroy matter. In fact, according to the Law of Conservation of Mass, formally discovered in the late 1700s, no matter can ever be created or destroyed. We can rearrange atoms, we can break their bonds, we can move them around, and we can release them into the air. But we cannot destroy them.
Today in class, students investigated this law. They measured the mass of a piece of paper, then crumpled that paper and weighed it again, then ripped up the paper and weighed it a third time. The mass of the paper never changed. Then they measured the mass of a match, burned it, and weighed it again. This time, the mass did change!
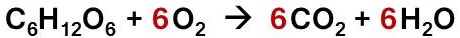
It turns out that the burning of wood generally follows the equation above (wood is a mixture, so it is hard to describe with one simple equation). Most of the wood that is burned turns into CO2 and H2O, and thus it escapes into the air. What is left behind are impurities, mostly charcoal and ash. Even though it seems as though burning wood breaks the law, it actually doesn’t. So rest easy everyone, you won’t go to jail today.