Today was the last day of our “Where’s the Evidence?” lab, and we saved the best for last. Today, students heated sugar (sucrose: C12H22O11) and observed the resulting chemical reaction. They piled the sugar onto a piece of tinfoil and held that foil directly over a burning candle. After a while, it began to melt and then react; the white sugar turned dark brown and seemed to release smoke into the air.
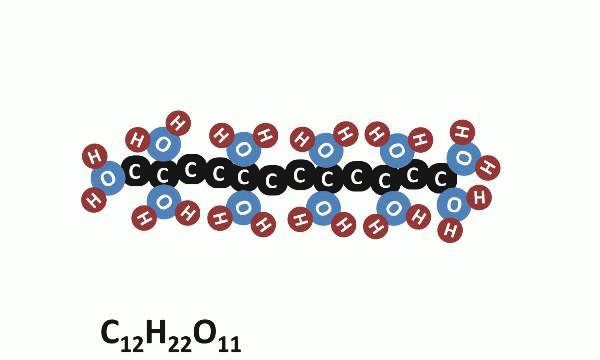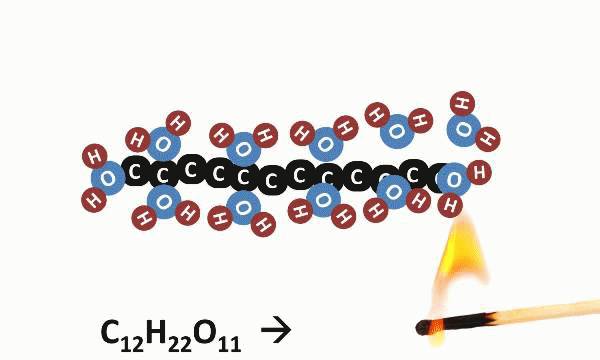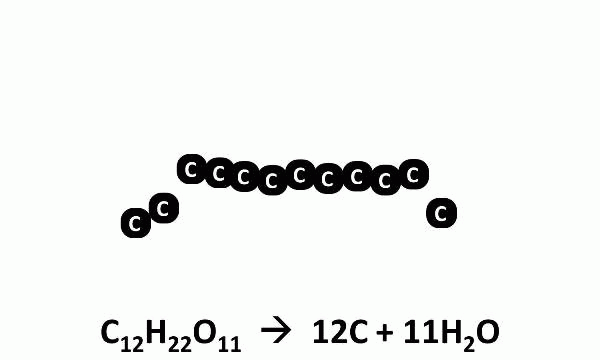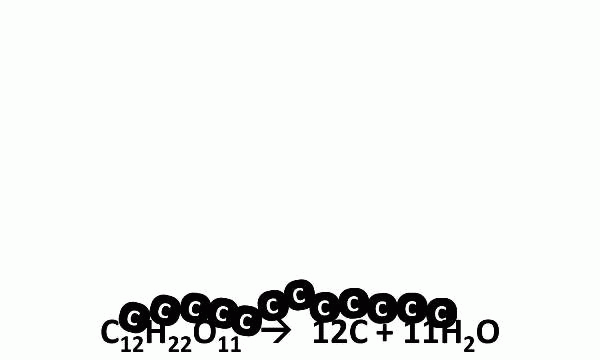
It turns out that the “smoke” was actually steam, the gaseous form of H2O. When heated, sugar molecules spontaneously decompose, and the black residue left behind is charcoal (carbon). Kind of sheds a new light on crème brulée, doesn’t it?
November 13 – Where’s the Evidence Day 4 (pg309)
November 13 – Day 4 Post-Lab (pg310)